 By doing that, scientists can decide the age of fossils using carbon-14. The remaining amount of carbon-14 can be measured and compared to the amount present in most living specimens. With that, carbon-14 starts to decay and becomes half of its initial amount in about 5730 years, which is called the half-life of carbon-14. When the organism dies, it stops taking carbon-14 sources. Organisms gain carbon-14 during photosynthesis or while eating organic matter. Carbon-14 decays to form nitrogen-14 atom. The decaying action of carbon-14 is spontaneous. Carbon-14 isotope makes up of about less than 0.01% of all naturally occurring carbon. Unlike other isotopes of carbon, carbon-14 is radioactive thus, it decays with time. The unit of Avogadro constant is mol -1.Ĭarbon-14 is the unstable isotope of carbon and contains 8 neutrons and 6 protons hence the mass number is 14. This number is defined as the Avogadro constant. 
An Italian Chemist, Amedeo Avogadro discovered this number in the nineteenth century. The exact number of atoms in 12 g of carbon-12 is found to be 6.02 x 10 23. The number of atoms in 12.00 g of carbon-12 is taken as the standard to define the mole. Just like the RAM, the mole is based on the carbon-12 isotope. The relative atomic mass (RAM) of elements is shown in the periodic table. Here, carbon-12 is taken as the standard atom. 
Carbon-12 atom is used to define the relative atomic mass scale, where the masses of other atoms are compared with the mass of an atom of the carbon-12 isotope. Therefore, it does not decay, unlike carbon-14. Carbon-12 isotope is stable and not radioactive. Carbon-12 atom contains 6 neutrons and 6 protons in its nucleus. Key Terms: Isotopes, Carbon, Carbon Isotopes, Mass Number, Neutrons, Carbon 12, Carbon 14, Radioactivity, Avogadro Constant What is Carbon 12Ĭarbon-12 isotope is the most abundant carbon isotope that makes up of about 98.89% of all naturally occurring carbon. What is the difference between Carbon 12 and Carbon 14 The main difference between carbon 12 and carbon 14 isotopes is their stability carbon 12 isotope is more stable than carbon 14. Hence, when considering the relative atomic mass of an element, it is usually assumed that the relative mass number is equal to the mass number of the major or abundantly existing isotope. Most elements with isotopes have one major isotope present abundantly in nature while the rest of the isotopes present in very small proportions. Likewise, carbon-13 isotope contains 7 neutrons and 6 protons, while carbon-14 isotope contains 8 neutrons and 6 protons. The mass number of carbon-12 is twelve as it contains 6 neutrons and 6 protons. For example, carbon is an element that exists in three forms in other words, carbon has three isotopes: carbon-12, carbon-13 and carbon-14. 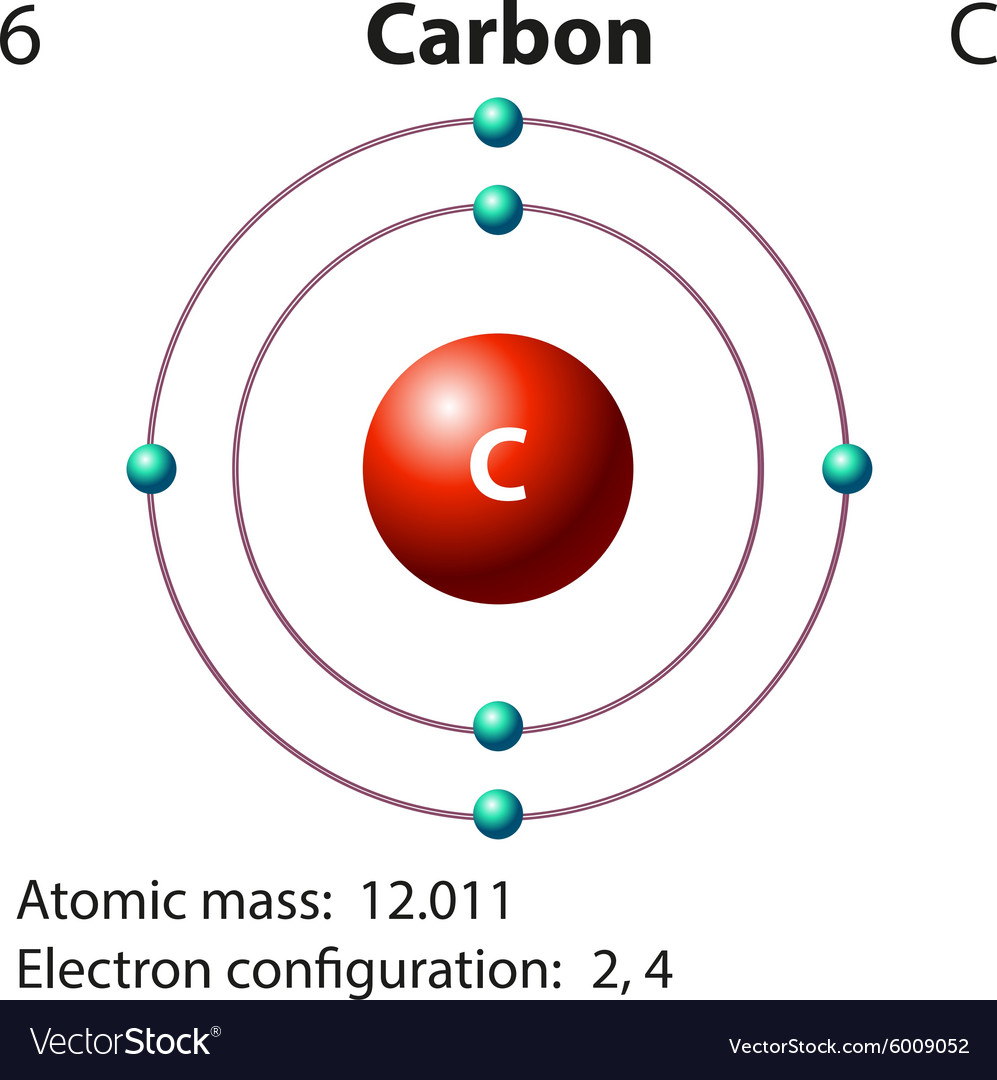 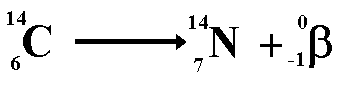
Thus, isotopes are denoted by their mass number. The mass number of an element is the sum of neutrons and protons in its nucleus. Hence, even if it has the same element, their mass is different. Isotopes of an element contain the same number of electrons and protons, but a different number of neutrons. Some elements can exist in different forms known as isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
